One Million Brain Cells and Counting
by Lindsay Borthwick
The amazing strides researchers have made in studying the brain
The Author
Science fiction writers would be hard-pressed to match the stories coming out of the Kavli neuroscience institutes these days. In February alone, came the report that a biologist and anthropologist had teamed up and created minibrains carrying an ancestral gene that could hold the key to what makes us human. Also, a physicist turned neuroscientist showed that it was possible to record more than 1 million neurons in the mouse cortex—a technological feat that takes researchers closer to creating a detailed, real-time motion picture of the circuits of the brain at work.
Evolution in a dish
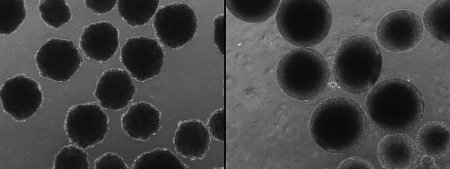
Alysson Muotri, PhD, has pioneered the use of stem cells to compare the brains of humans and our closest living relatives, chimps and bonobos. Now, in a proof-of-concept study published this month in Science, researchers led by Muotri and UC San Diego anthropologist Katerina Semendeferi have gone a step further: They “Neanderthaliz-ed” brain organoids, three-dimensional clusters of brain cells also known as “mini brains.” The organoids were grown in a lab from human stem cells that had been modified to contain a Neanderthal version of a gene, NOVA1, which is thought to have played an important role in the evolution of modern humans. Compared to modern human brain organoids, the Neanderthal-ized ones were strikingly different in shape and function, including at synapses, the junctions between neurons. Muotri and Semendeferi plan to continue studying the impact of NOVA1 on the mini brains, as well as dozens of other genes that differ between modern and extinct humans. Both researchers are affiliated with the Kavli Institute for Brain and Mind at UC San Diego.
Valuable Noise
A recent article in Quanta magazine on the brain’s “background noise” highlights the research of UC San Diego cognitive scientist Bradley Voytek, who is also affiliated with the Kavli Institute for Brain and Mind. He is part of a growing number of researchers who views the brain’s electrical noise as a potential treasure trove of information about its inner workings. He’s developing computational tools to help neuroscientists separate regular oscillations from aperiodic brain activity and look for meaningful signals in the “leftover” electrical noise.
Developmental Checkpoint
What triggers an immature brain cell to stop multiplying and instead differentiate into a mature neuron? That question is central to understanding brain development and the precise orchestration that leads to the elaborately structured mammalian brain. “To my mind, how these decisions get made is one of the most fundamental questions in stem cell biology,” says Georgia Panagiotakos, PhD, a member of the Kavli Institute for Fundamental Neuroscience at the University of California, San Francisco, whose research team recently identified a protein complex that plays a critical role in triggering differentiation. The team showed that a protein complex in progenitor cells known as BAF swaps several of its subunits, triggering large-scale changes in gene expression that drive cells to differentiate. Panagiotakos showed that without the BAF subunit switch, cells stay stuck in the progenitor state. Several of the genes encoding the protein complex are altered in disorders such as autism and intellectual disability, suggesting it plays an important role in healthy brain development.
Birds vs. Mammals
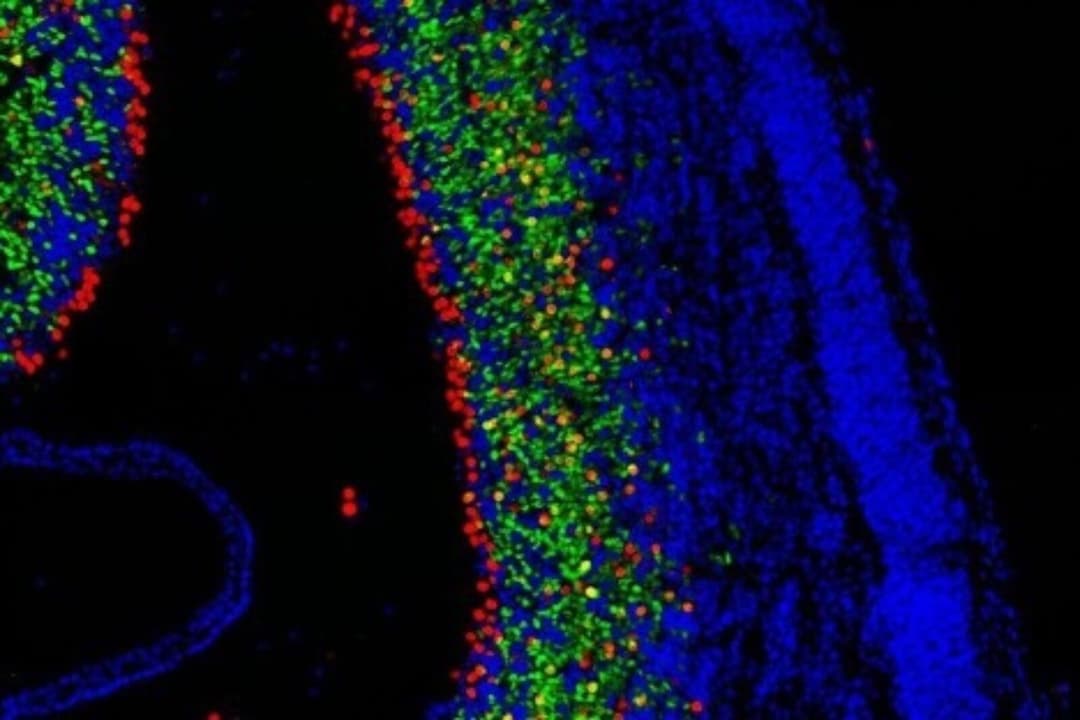
Also out of the Kavli Institute for Fundamental Neuroscience (Kavli IFN) is an in-depth look at the major classes of neurons that comprise the song-control system in birds. The study, led by Kavli IFN member Michael Brainard, PhD, and published in Science, enables a comparison of the brain organization in mammals and birds, especially between a cortex-like structure in the avian forebrain and the neocortex in mammals. The new findings suggest the two structures have different evolutionary origins. However, excitatory neurons in the two animals share overlapping transcriptional profiles, which “may account for the evolution of similar complex motor and cognitive abilities in mammals and birds, including vocal learning,” according to the authors.
Hitting a Million
Rockefeller University’s Alipasha Vaziri has simultaneously recorded the activity of more than 1 million neurons across the mouse cortex. Vaziri and his team broke the 1-million-neuron barrier using a new high-speed optical imaging technique, Light Beads Microscopy. Compared to existing techniques, it scales the total number of neurons recorded by 3 orders of magnitude, making it possible to study how large, dynamic populations of cortical neurons encode and process sensory and behavioral information. Vaziri is associate director of Rockefeller’s Kavli Neural Systems Institute. He reported the new research, which has not been peer-reviewed, in a preprint posted to bioRxiv.
Excited to share our new manuscript showing volumetric Ca #imaging of 1 million neurons across the mouse cortex at cellular resolution using Light Beads #Microscopy (LBM). https://t.co/EISFbPJSkT Congratulations to the entire team! #neurocience #optics #neurotechnology #photonics pic.twitter.com/Deflsbu0H3
— Vaziri Lab (@VaziriLab) February 23, 2021
Sound of Silence
A new study illuminates how the brain responds to sound at the very earliest stages of life. Patrick Kanold, PhD, an affiliate of the Kavli Neuroscience Discovery Institute at Johns Hopkins University, previously showed that subplate neurons—transient neurons that are some of the first to form during brain development—play a key role in cortical development and plasticity, including in the brain’s sound-processing circuits. In the new study he wondered, what sound signals do the neurons receive, and what—if anything—happens in response to such signals? Working with mice, Kanold’s team showed that sound exposure altered circuits between subplate neurons and the cortex even before the animals’ ear canals had opened. “In these mice we see that the difference in early sound experience leaves a trace in the brain, and this exposure to sound may be important for neurodevelopment,” says Kanold.
Early Adopters
Edvard Moser has called on European Union officials not to undervalue early-stage research. Moser is founding director of the Kavli Institute for Systems Neuroscience and co-director of the Centre for Neural Computation at the Norwegian University of Science and Technology.
None of this could have happened if there hadn’t been a mountain of basic research in front of the pandemic. All of this builds on a huge amount of knowledge.
Edvard Moser