A Flash of Light Changes Cell Activity - and Understanding of Disease
(Originally published by Yale University)
July 30, 2012
With a milliseconds-long flash of blue light, Yale University researchers regulated a critical type of signaling molecule within cell membranes, another illustration of the power of light-based techniques to manipulate cell functions and thus to study mechanisms of disease.

One of the most exciting new research approaches of recent years is called optogenetics or the use of genetically encoded probes to make cell functions sensitive to light. The new study, published the week of July 30 in the journal Proceedings of the National Academy of Sciences, is one of the first to use light to regulate metabolic activity in the membranes of cells.
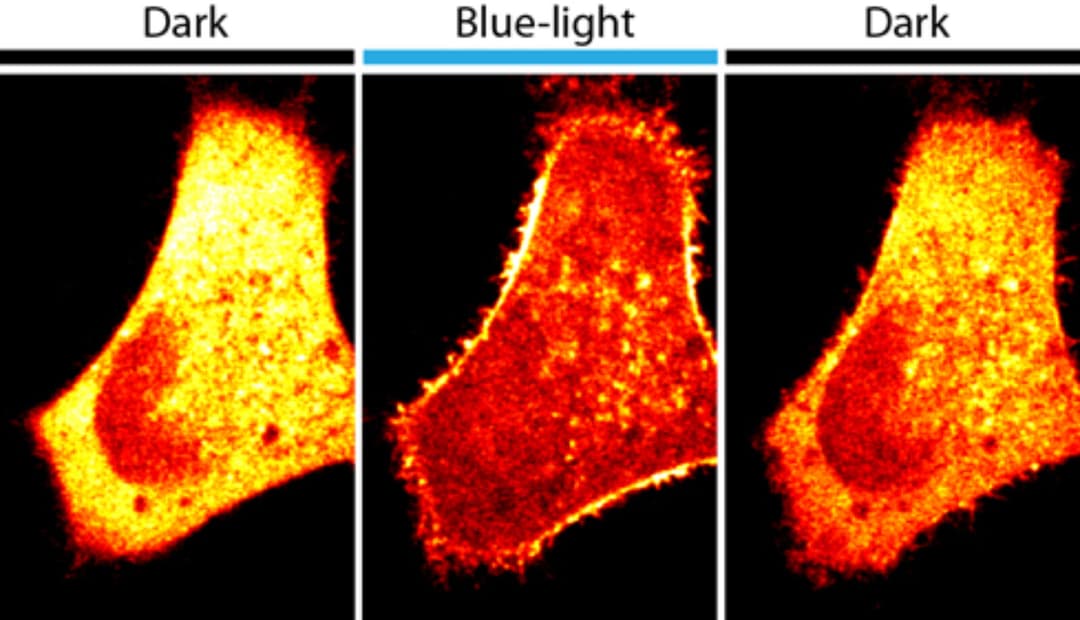
"The cell outer membrane is the part of the cell that communicates with the environment,” said Olof Idevall-Hagren, postdoctoral researcher and lead author of the paper. “Many cellular processes start at the cell outer membrane.”
The team combined a plant protein that is sensitive to blue light with enzymes that catalyze the metabolism of signaling lipids within the cell membranes. When the complex was expressed in animal cells, scientists changed properties of cells such as their shape or ability to move simply by using blue light. By turning off the light, the researchers were able to rapidly reverse the changes they induced. They were also able to regulate activities within a region of a cell by illuminating the area.
“This is a powerful tool to acutely manipulate the metabolism of membrane lipids and to study the resulting changes of cell behavior in real time,’ said Pietro De Camilli, the Eugene Higgins Professor of Cell Biology, investigator for the Howard Hughes Medical Institute and member of the Kavli Institute for Neuroscience at Yale University. “Abnormal metabolism of lipids in cell membranes has been implicated in many diseases, such cancer, diabetes and neurodegeneration, including Alzheimer’s Disease.”
The team, whose primary focus is the function of the nervous system, hopes to use the technology to manipulate lipids in specific parts of neurons to investigate how the disruption of these mechanisms contribute to the development of disease.
Other authors of the study are Eamonn J. Dickson and Bertil Hille of the University of Washington, and Derek K. Toomre of Yale.
The study was funded by the National Institutes of Health, the Ellison and Simons Foundations and the Swedish Research Council.